Cryptomonads = Goniomonas + plastid-containing cryptophytes
Cryptophyta
Kerstin Hoef-Emden and John M. Archibald


This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Cryptomonads are aquatic unicellular eukaryotes that inhabit both marine and freshwater environments. Most cryptomonads are photosynthetic (and are thus referred to as cryptophytes) and possess plastids that are very diverse in pigmentation (Figure 1). In addition to being important from an ecological perspective, the cryptophytes are of pivotal significance in terms of our understanding of endosymbiosis and the evolution of plastids. This is because cryptophytes acquired photosynthesis by the process of secondary (i.e., eukaryote-eukaryote) endosymbiosis, having engulfed and assimilated a red algal endosymbiont at some time during their evolutionary past (Archibald and Keeling 2002; Bhattacharya et al. 2003; Gould et al. 2008). As a result, cryptophytes are extremely complex from a genetic and cell biological perspective, having a four-membrane-bound plastid and four distinct DNA-containing cellular compartments, the plastid, mitochondrion, host nucleus and endosymbiont nucleus, the latter being referred to as the nucleomorph. The cryptophyte nucleomorph genome is highly reduced in structure and coding capacity, and is the focus of ongoing research aimed at understanding the pattern and process of secondary endosymbiosis (Gilson and McFadden 2002; Archibald 2007).
Characteristics
Cryptomonads are unicellular biflagellate cells, most of which are photosynthetic (= cryptophytes). They are characterized by the following general features:
- Asymmetric cell shape
- Cell invagination at the ventral side lined with ejectosomes (explosive organelles)
- Flagella insert with a shift to the right side in the vestibulum of the cell invagination
- Sandwich-layered cell envelope (the periplast) consisting of proteinaceous inner and outer periplast components with the plasma membrane in between (Figure 2)
- Plastid surrounded by four membranes, the outer membrane of which is continuous with the nuclear envelope and rough endoplasmic reticulum
- Starch grains, 80S ribosomes and a highly reduced second nucleus—the nucleomorph—in the periplastidial space between the two outer and the two inner plastid membranes
- Lamellar arrangement of thylakoids in plastid
- Two different light harvesting complexes: one type with chlorophylls a and c2 on the outside of the thylakoids and another type consisting of highly modified biliprotein in the thylakoid lumina, no phycobilisomes
- Additional pigments: predominantly alloxanthin
- Storage substances: starch in the periplastidial space, oil droplets in the cytoplasm
- Mitochondria with flat cristae
Only one aplastidic cryptomonad genus is known, Goniomonas (Mignot 1965). It differs in cell shape, cell invagination and flagellar structures from the photosynthetic genera. Within the photosynthetic genus Cryptomonas, three lineages lost photosynthesis independently (Hoef-Emden 2005). These colorless cryptophytes still contain leucoplasts (i.e., colorless plastids) and nucleomorphs.
A few cryptophyte genera have been shown to possess a dimorphic life history, a fact that has complicated efforts to produce reliable taxonomic schemes (see below). Very little direct evidence for the presence of two different morphotypes currently exists: clear examples have been observed only in a few clonal cultures of the genera Proteomonas and Cryptomonas (Hill and Wetherbee 1986; Hoef-Emden and Melkonian 2003). In these cases, the morphotypes shared the same type of biliprotein but differed in periplast type, cell shape, flagellar apparatus ultrastructure and sometimes in cell size. Indirect evidence for dimorphism in other genera has come from consideration of phylogenetic trees (Rhinomonas/Rhodomonas/Storeatula clade and Geminigera/Plagioselmis/Teleaulax clade). The pervasiveness of this characteristic across the full spectrum of cryptophyte diversity is unknown, though it is possible that a dimorphic life history is an ancient characteristic of the group, inherited from a common cryptophyte ancestor (Hoef-Emden et al. 2002).


Figure 2. Morphology of the cryptophytes. © 2008 Kerstin Hoef-Emden
Morphology of the Cryptomonads
Cryptophytes can be easily identified by light microscopy due to their asymmetrical cell shapes and their red, olive, brown, blue or blue-green plastids (Figure 2). The apex is often shifted to the left side of the cells, whereas the two unequal flagella insert in the vestibulum of a cell invagination that is shifted to the right. The cell invagination is not always clearly visible, but the large ejectosomes, explosive organelles lining the furrow-gullet system, often are discernible (e.g., Skuja 1948; Huber-Pestalozzi 1950; Butcher 1967). The asymmetric insertion of the flagella causes an unbalanced rotating around the longitudinal axis while swimming. The larger the cells and the more the cells are dorsally or ventrally bent, the more expressed is this “swaying”. The nucleus is always found in the antapical half of the cells. The parietal plastid may be deeply bi-lobed and H-shaped. Sometimes two plastids are found. A single pyrenoid may be present centrally in the plastid, towards the dorsal side of the cell. In bi-lobed plastids this position corresponds to the connection between the two plastid lobes. In some species, two or more pyrenoids are closely appressed to the left and right inner sides of the plastid lobes. In all freshwater species, a contractile vacuole for regulation of osmotic pressure is found in the apex of the cell.
Cryptophytes share several unique features that are discernible only by electron microscopy. The cryptophyte-specific cell surrounding, termed the periplast, is a sandwich-layered structure consisting of proteinaceous inner (IPC) and surface periplast components (SPC) that embrace the plasma membrane (Figure 2; Kugrens et al. 1986; Brett et al. 1994). Small light microscopically invisible ejectosomes are scattered in a regular pattern underneath the periplast. Both types of ejectosomes, the large furrow-gullet ejectosomes and the small periplast ejectosomes, consist of two connected ribbons tightly coiled like springs (Morrall and Greenwood 1980). They become abruptly discharged upon mechanical or chemical irritation causing the cells to shoot away from the irritant.
The cryptophyte plastid is surrounded by four membranes and harbors a second highly reduced eukaryotic genome, the nucleomorph (see Cell Biology), in its periplastidial compartment between the two outer and the two inner membranes. The unique highly modified biliproteins are not found on the outside of the thylakoids, but as electron-dense material in the thylakoid lumina (Gantt et al. 1971).
The flagella of cryptophytes resemble those of the stramenopiles. Two rows of stiff flagellar hairs on the longer locomotoric flagellum cause a reversal of thrust. With the flagellum pointing in the direction of swimming, the cells are pulled behind. The shorter flagellum bears only one row of flagellar hairs. Several variations of this pattern have been reported by Kugrens et al. (1987). The flagellar hairs of the locomotoric and trailing flagella are attached in a different way and also differ in length and in number of terminal filaments (Hibberd et al. 1971). In the aplastidic cryptomonad Goniomonas, the appendages of its flagella are highly modified. In addition to fine filaments, one flagellum is covered with a row of curved spikes that probably impede a cryptophyte-like swimming behaviour (Kugrens et al. 1987).
Habitat and Ecology
Cryptophytes successfully conquered freshwater, brackish and marine habitats (Klaveness 1988). They have even been found in snow (Javornický and Hindák 1970). As non-toxic plankters, cryptophytes serve as important prey for diverse predators and are used as food in fish farming (Pedrós-Alió et al. 1995; Brown et al. 1997; Pastoureaud et al. 2003; Tirok and Gaedke 2007). A greater diversity of genera is found in marine habitats, whereas the densest populations of cryptophytes often develop in the deep-water layers of freshwater lakes (genus Cryptomonas; Pedrós-Alió et al. 1995; Gervais 1997, 1998). Due to their biliprotein light harvesting complexes, cryptophytes can photosynthesize in low light conditions (Gervais 1997; Hammer et al. 2002). Cryptomonas species migrate in a daily rhythm in the water column of freshwater lakes from the chemocline close to the anaerobic bottom layer up to the epilimnion (Pedrós-Alió et al. 1995; Gervais 1997, 1998). It has been hypothesized that this migration may either be a strategy to circumvent contact with predators or a way for the Cryptomonas cells to take advantage of the nutrients available in deep waters and then return to the nutrient-depleted but light-flooded epilimnion for photosynthesis (Salonen et al. 1984; Klaveness 1988; Pedrós-Alió et al. 1995; Camacho et al. 2001). The formation of palmella, flocks of flagellated cells embedded in mucus, probably serves as a protection against predators. Cryptomonas cells, and perhaps also other cryptophytes, can produce globular thick-walled cysts as resting stages to survive unfavorable environmental conditions (Lichtlé 1979, 1980).
The phagotrophic cryptomonad genus Goniomonas does not swim in the water column, but glides along substrates in search for prey. Bacterivory has been reported also from the plastid-containing cryptophytes, and endocytic bacteria have been found in ultra-thin sections (Tranvik et al. 1989; Kugrens and Lee 1990; Schnepf and Melkonian 1990; Marshall and Laybourn-Parry 2002). The bacteria found in the cells apparently entered the cells via the contractile vacuole (Kugrens and Lee 1990, 1991). Different from Goniomonas, a cytostome or cytopharynx is not present in plastid-containing cryptophytes.
Cell Biology

Figure 3. The four genome-containing compartments of plastid-bearing cryptophytes. © 2008 John M. Archibald
Cryptophytes acquired photosynthesis by secondary endosymbiosis, and their plastids possess four membranes (Figure 3). The inner two membranes correspond to the inner and outer membranes of the cyanobacterial progenitor of the plastid, while the third and fourth (outermost) membranes appear to be derived from the plasma membrane of the secondary endosymbiont and the phagocytic vacuole of the host eukaryote, respectively. The space between the inner and outer pairs of plastid membranes, which corresponds to the remnant cytoplasm of the engulfed algal cell, is known as the periplastidial space and contains the nucleomorph. Little is known about the cellular activities taking place in this compartment and in the nucleomorph itself, beyond starch synthesis/storage and core biochemical processes such as DNA replication, transcription and translation. The outermost plastid membrane, which is studded with ribosomes, is continuous with the nuclear envelope and endoplasmic reticulum (ER). This arrangement has been referred to as the chloroplast ER (Gibbs 1981) and results in a close association between the plastid-nucleomorph complex and the host cell nucleus.
The process of targeting host nucleus-encoded proteins to the plastid is considerably more complex in cryptophytes than in plants and algae, whose plastids are of primary endosymbiotic origin. In cryptophytes and other secondary plastid-containing organisms, plastid-targeted proteins possess a bipartite amino-terminal leader sequence consisting of a signal peptide followed by a transit peptide. Together, these topogenic signals allow newly translated proteins across as many as four membranes to their site of function (Gould et al. 2008). It has been hypothesized that the nucleomorph-encoded ER-associated degradation (ERAD) machinery of the endosymbiont has been co-opted to perform a new function. Instead of exporting proteins for degradation from the non-existent periplastidial ER, it may now be involved in importing nucleus-encoded proteins into the periplastidial space or plastid (Sommer et al. 2007).
Two types of light harvesting complexes are found in the cryptophyte plastid, a chlorophyll a/c2 complex on the outside of the thylakoid membranes and a biliprotein that, peculiarly, accumulates as dense material in the thylakoid lumina (Gantt et al. 1971; Vesk et al. 1992). In red algae, the biliproteins are components of very large light harvesting complexes termed phycobilisomes, which are attached to the exterior of the thylakoids. At least three different types of biliproteins organized in heterohexa- or heterodocameric discs contribute to energy transfer (Gantt 1996). In the cryptophyte plastid, only one highly modified biliprotein has been retained, whereas the rest of the phycobilisome has disappeared during the course of evolution (Hill and Rowan 1989). The quarternary structure of the biliprotein changed from a heterohexa- or heterodocameric disc to an αα′ββ heterodimer (Wilk et al. 1999). The β subunits of all sequenced cryptophyte biliproteins grouped in phylogenetic analysis with red algal phycoerythrins, irrespective of a blue (“phycocyanin”) or red (phycoerythrin) color, indicating that all known types of cryptophyte biliproteins evolved from a red algal phycoerythrin. Exchanges of the covalently bound linear tetrapyrroles gave rise to different colors and absorption spectra (Glazer and Wedemayer 1995). The a subunits of the biliproteins are so highly diverged that their evolutionary origin could not be deduced (Apt et al. 1995). Whereas in red algae, all genes for biliprotein subunits are found in the plastid genome, the α subunits of the cryptophyte biliprotein are encoded in the nucleus (Jenkins et al. 1990; Broughton et al. 2006). Of all subunits, only one of the two slightly different α subunits contains a targeting sequence for import into the thylakoid lumen, possibly via the TAT pathway (Reith and Douglas 1990; Broughton et al. 2006; Gould et al. 2007). It has therefore been hypothesized that the biliprotein is assembled in the plastid stroma, and then the complete complex imported into the thylakoid lumen guided by the targeting sequence of the α1 subunit (Broughton et al. 2006). Despite its extreme divergence and peculiar locality, the cryptophyte biliprotein is a fully functional light-harvesting complex, in low light conditions or under green light (Lichtlé et al. 1980; Doust et al. 2006).
The Nucleomorph and its Genome
Together with the chlorarachniophytes, the cryptophytes are unusual in that they possess a nucleomorph, a small, double-membrane-bound organelle residing between the inner and outer sets of plastid membranes (Figure 2). The nucleomorph was first described in the 1970s (Greenwood 1974; Greenwood et al. 1977) and has been studied using cytochemistry and, more recently, molecular biology. Phylogenetic and comparative genomic analyses (e.g., Douglas et al. 1991; Douglas et al. 2001; Hoef-Emden et al. 2002) have shown convincingly that the cryptophyte nucleomorph is a remnant nucleus of red algal origin. The structure and function of the cryptophyte nucleomorph genomes has provided fascinating insight into the process of genome reduction and compaction.
Preliminary karyotypic analysis of the cryptophyte Pyrenomonas salina (now Rhodomonas salina) revealed that its nucleomorph genome is ~660 kilobase-pairs (Kbp) in size and comprised of only three chromosomes (~195, 225 and 240 Kbp; Eschbach et al. 1991; Maier et al. 1991). As of 2008, the known size distribution of cryptophyte nucleomorph genomes is between ~450 and ~850 kilobase-pairs (Kbp) (Rensing et al. 1994; Lane et al. 2006; Archibald 2007), presumably orders of magnitude smaller than the red algal genomes from which they evolved. The presence of three nucleomorph chromosomes (Figure 4) appears to be a universal feature that, intriguingly, is shared with the nucleomorphs of an unrelated algal group, the chlorarachniophytes.

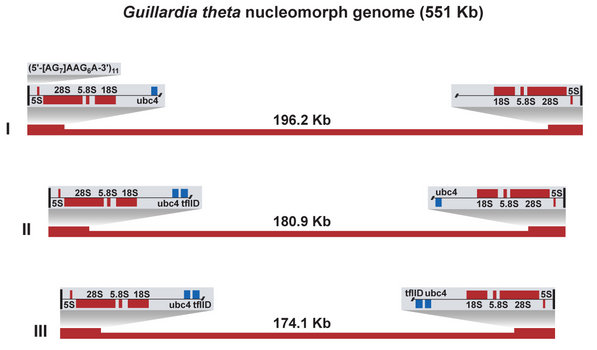
Figure 4. The three nucleomorph chromosomes of Guillardia theta. © 2008 John M. Archibald
The first nucleomorph genome to be sequenced was that of the model cryptophyte Guillardia theta (Douglas et al. 2001). The G. theta nucleomorph genome is 551 kilobase-pairs (Kbp) in size, partitioned amongst three chromosomes of ~196, 181 and 174 Kbp (Figure 4). The chromosomes are characterized by the presence of near-identical sub-telomeric ribosomal DNA repeats. The telomeres themselves are unusual in structure, being comprised of a (5’-[AG]7AAG6A-3’)11 repeat. The overall A,T content of the genome is high, ~75%. The genome possesses genes primarily with ‘housekeeping’ functions such as protein folding/degradation, transcription, and translation. 30 genes encoding plastid-targeted proteins are present. In 2007, the nucleomorph genome of another cryptophyte, Hemiselmis andersenii, was sequenced (Lane et al. 2007). At ~572 Kbp, the H. andersenii genome is slightly larger than that of G. theta. The overlap in gene compliment between the two genomes is extensive (H. andersenii contains the identical set of 30 plastid protein genes), as is the degree of gene order conservation (synteny). The most striking difference between the two genomes is the complete absence of spliceosomal introns and genes for splicing proteins and RNAs in the H. andersenii genome. The G. theta genome contains 17 introns between 42 and 52 base pairs in size. The gene density in the cryptophyte nucleomorph genome is high (~1 Kbp/gene), a fact that has important implications for the process of transcription. Numerous instances of mRNAs encoding more than one gene have been discovered in the G. theta nucleomorph (Williams et al. 2005), presumably a result of the transcriptional regulatory elements being located within or beyond neighbouring genes.
Relationships of Cryptomonads to Other Organisms
The evolutionary position of the host cell component of cryptophytes on the eukaryotic tree is not well understood. Cryptomonads were once suggested to belong to the Chromista, a tripartite lineage comprised of the cryptomonads, haptophytes and photosynthetic stramenopiles (Cavalier-Smith 1986). These three lineages each possess a secondary plastid of red algal origin, and it has been hypothesized that this organelle originated in a single endosymbiotic event in their common ancestor, and possibly much earlier, in a common ancestor shared by chromists and alveolates, which are comprised of dinoflagellates, apicomplexans and ciliates. It is not clear whether ‘chromalveolates’ (chromists , alveolates) represent a monophyletic lineage and, if so, whether the red algal plastids of photosynthetic chromalveolates are in fact the product of a single secondary endosymbiosis. Molecular phylogenetic analyses of plastid-, mitochondrion- and nucleus-encoded genes have produced inconclusive results (e.g., Yoon et al. 2002; Harper et al. 2005; Khan et al. 2007; Sanchez-Puerta et al. 2007). Multigene phylogenies of nuclear loci suggest that while cryptomonads and haptophytes are specifically related, the stramenopiles are not their closest relatives (Burki et al. 2007; Hackett et al. 2007; Patron et al. 2007; Burkiet al. 2008). Possible evolutionary connections between cryptomonads and several newly discovered protist lineages have also been established, including the katablepharids (Okamoto and Inouye 2005), telonemids (Shalchian-Tabrizi et al. 2006) and picobiliphytes (Not et al. 2007).
History of Systematics
The Age of Light Microscopy
The use of the prefix “crypto” in names of higher classification ranks (division Cryptophyta, class Cryptophyceae, order Cryptomonadales, family Cryptomonadaceae, cryptomonad, cryptophyte) originates from the first described cryptophyte genus Cryptomonas described by Ehrenberg (1831).
Prior to the use of electron microscopy, light microscopical examinations of cryptophyte cells from field samples were state-of-the-art. Several genera and around 200 cryptophyte species have been described based on morphological characters visible by light microscopy (Huber-Pestalozzi 1950, Butcher 1967, Starmach 1974). Species descriptions included to varying degrees cell size, cell color, cell shape, number of ejectosome rows, presence and number of Maupas ovals, presence and number of pyrenoids, number and shape of plastids, and type of cell invagination. The morphological species concept was based on the assumption that different morphological characters indicate different species. Pringsheim (1968), however, stressed the necessity to test this assumption by examining the reliability and specificity of morphological characters using clonal cultures instead of field material. After examining many Cryptomonas strains, he came to the conclusion that it was not possible to apply the morphological species concept to Cryptomonas due to a lack of distinctive characters and too many transitional forms (Pringsheim 1968).
The Age of Electron Microscopy
Cryptophyte strains have been examined by transmission and scanning electron microscopical methods. These studies unveiled a considerable variation of delicate periplast structures (Figure 5), different shapes of cell invagination and differences in flagellar roots. The nucleomorph could be found in different positions in the cell (free in the periplastidial space, embedded in the nucleus or embedded in the pyrenoid matrix). These findings resulted in descriptions of new cryptophyte genera based on morphological characters visible by electron microscopy only (summarized in Clay et al. 1999). Different from the morphological characters used to delimit species by light microscopy, these ultrastructural features were clearly distinctive. Except for one strain expressing two different morphotypes in a culture (Hill and Wetherbee 1986), the type of periplast, structure of the flagellar root system, and also position of the nucleomorph seemed to be stable characters in a culture.


Figure 5. Inner periplast component made of distinct polygonal plates (left, in detail) and a sheet-like periplast (right). © 2008 Kerstin Hoef-Emden
Spectrophotometry
In the 1950s, the first spectrophotometric analyses of cryptophytes were performed and provided evidence that each strain contains only one type of biliprotein (summarized by Hill and Rowan 1989). Thus far, eight different types of biliprotein have been found, each one stable in a clonal culture (Figure 6; Hill and Rowan 1989; Hoef-Emden 2008).


Figure 6. Cryptophyte biliproteins. From left to right: PC630, PC577, PE555, PE566. © 2008 Kerstin Hoef-Emden
In sum, although cryptophyte ultrastructural characters and their biliproteins were stable in most cultures, phylogenetic analyses performed thus far have revealed that their use as diagnostic characters does not necessarily result in a natural system.
Discussion of Phylogenetic Relationships
Inconsistencies in the systematics of plastid-containing cryptophytes have become apparent in molecular phylogenies inferred from DNA sequences of nucleus and/or nucleomorph ribosomal operons (Marin et al. 1998; Deane et al. 2002; Hoef-Emden et al. 2002). In all phylogenetic trees, the photosynthetic cryptophytes are subdivided into five clades and two single-strain lineages (Figure 7):
- Cryptomonas
- Chroomonas/Komma and Hemiselmis genus complex
- Rhodomonas/Rhinomonas/Storeatula genus complex
- Guillardia/Hanusia clade
- Geminigera/Plagioselmis/Teleaulax genus complex
- Proteomonas sulcata
- Falcomonas daucoides
Several of the clades do not correspond to genera, and not all morphologically defined genera form distinct sub-groups within these clades (Marin et al. 1998; Deane et al. 2002; Hoef-Emden et al. 2002). Only the freshwater genus Cryptomonas and the genus Hemiselmis have been examined in greater detail and revised so far. Thus, cryptophyte systematics is in a state of flux.
Other Names for Cryptomonads = Goniomonas + plastid-containing cryptophytes
- Cryptophyta
- Cryptomonadida
- Cryptomonadidae
- Cryptophytes
- Cryptophyceae
- Cryptomonadales
- Cryptomonadaceae
References
Apt KE, Collier JL, Grossman AR (1995) Evolution of the phycobiliproteins. J. Mol. Biol. 248: 79-96
Archibald JM (2007) Nucleomorph genomes: structure, function, origin and evolution. BioEssays 29: 392-402
Archibald JM, Keeling PJ (2002) Recycled plastids: a green movement in eukaryotic evolution. Trends Genet. 18: 577-584
Bhattacharya D, Yoon HS, Hackett JD (2003) Photosynthetic eukaryotes unite: Endosymbiosis connects the dots. BioEssays 26: 50-60
Brett SJ, Perasso L, Wetherbee R (1994) Structure and development of the cryptomonad periplast. Protoplasma 181: 106-122
Broughton MJ, Howe CJ, Hiller RG (2006) Distinctive organization of genes for light-harvesting proteins in the cryptophyte alga Rhodomonas. Gene 369: 72-79
Brown MR, Jeffrey SW, Volkman JK, Dunstan GA (1997) Nutritional properties of microalgae for mariculture. Aquaculture 151: 315-331
Burki F, Shalchian-Tabrizi, K Minge, M Skjaeveland, Å Nikolaev SI, Jakobsen KS, Pawlowski J (2007) Phylogenomics reshuffles the eukaryotic supergroups. PLoS One 8: e790
Burki F, Shalchian-Tabrizi K, Pawlowski J (2008) Phylogenomics reveals a new 'megagroup' including most photosynthetic eukaryotes. Biology Letters doi:10.1098/rsbl.2008.0224
Butcher RW (1967) An Introductory Account of the Smaller Algae of British Coastal Waters. Part IV: Cryptophyceae. Fishery Investigations, Series IV. Ministry of Agriculture, Fisheries & Food, Her Majesty's Stationary Office, London
Camacho A, Vicente E, Miracle MR (2001) Ecology of Crypomonas at the chemocline of a karstic sulfate-rich lake. Mar. Freshwater Res. 52: 805-815
Cavalier-Smith T (1986) The kingdom Chromista: Origin and Systematics. Progr. Phycol. Res. 4: 309-347
Clay B, Kugrens P (1999). Characterization of Hemiselmis amylosa sp. nov. and phylogenetic placement of the blue-green cryptomonads H. amylosa and Falcomonas daucoides. Protist 150: 297-310
Clay B, Kugrens P, Lee RE (1999) A revised classification of the Cryptophyta. Bot. J. Linn. Society 131: 131-151
Deane JA, Hill DRA, Brett SJ, McFadden GI (1998) Hanusia phi gen. et sp. nov. (Cryptophyceae): characterization of 'Cryptomonas sp. φ'. Eur. J. Phycol. 33: 149-154
Deane JA, Hill DRA, Brett SJ, McFadden GI (2002) Cryptomonad evolution: Nuclear 18S rDNA phylogeny versus cell morphology and pigmentation. J. Phycol. 38: 1236-1244
Douglas SE, Murphy CA, Spencer DF, Gray MW (1991) Cryptomonad algae are evolutionary chimaeras of two phylogenetically distinct unicellular eukaryotes. Nature 350: 148-151
Douglas SE, Penny SL (1999) The plastid genome of the cryptophyte alga, Guillardia theta: Complete sequence and conserved synteny groups confirm its common ancestry with red algae. J. Mol. Evol. 48: 236-244
Douglas SE, Zauner S, Fraunholz M, Beaton M, Penny S, Deng LT, Wu X, Reith M, Cavalier-Smith T, Maier UG (2001) The highly reduced genome of an enslaved algal nucleus. Nature 410: 1090-1096
Doust AB, Wilk KE, Curmi PMG, Scholes GD (2006) The photophysics of cryptophyte light-harvesting. J. Photochem. Photobiol. A 184: 1-17
Ehrenberg CG (1831) Symbolae physicae seu Icones et Descriptiones Animalium Evertebratorum sepositis Insectis quae ex itinere per Africanum borealem et Asiam occidentalem Friderici Guilelmi Hemprich et Christiani Godofredi Ehrenberg medicinae et chirurgiae doctorum studio novae aut illustratae redierunt. Mittler, Berlin
Ehrenberg CG (1832) Über die Entwickelung und Lebensdauer der Infusionsthiere; nebst ferneren Beiträgen zu einer Vergleichung ihrer organischen Systeme. Abh. Königl. Akad. Wiss. Berlin, Physik. Klasse 1831: 1-154
Ehrenberg CG (1838) Die Infusionsthiere als vollkommene Organismen: Ein Blick in das tiefere organische Leben der Natur. Nebst einem Atlas von colorirten Kupfertafeln. Voss, Leipzig
Eschbach S, Hofmann CJ, Maier UG, Sitte P, Hansmann P (1991) A eukaryotic genome of 660 kb: electrophoretic karyotype of nucleomorph and cell nucleus of the cryptomonad alga, Pyrenomonas salina. Nucleic Acids Res. 19: 1779-1781
Fresenius G (1858) Beiträge zur Kenntniss mikroskopischer Organismen. Abh. Senckenberg. Naturf. Ges. 2: 211-242
Gantt E (1996) Pigment protein complexes and the concept of the photosynthetic unit: Chlorophyll complexes and phycobilisomes. Photosynth. Res. 48: 47-53
Gantt E, Edwards MR, Provasoli L (1971) Chloroplast structure of the Cryptophyceae. J. Cell Biol. 48: 280-290
Gervais F (1997). Diel vertical migration of Cryptomonas and Chromatium in the deep chlorophyll maximum of a eutrophic lake. J. Plankton Res. 19: 533-550
Gervais F (1998). Ecology of cryptophytes coexisting near a freshwater chemocline. Freshw. Biol. 39: 61-78
Gibbs SP (1981) The chloroplast endoplasmic reticulum: structure, function, and evolutionary significance. Int. Rev. Cytol. 72: 49-99
Gilson PR, McFadden GI (2002) Jam packed genomes--a preliminary, comparative analysis of nucleomorphs. Genetica 115: 13-28
Gillott MA, Gibbs SP (1983) Comparison of the flagellar rootlets and periplast in two marine cryptophytes. Can. J. Bot. 61: 1964-1978
Glazer AN, Wedemayer GJ (1995) Cryptomonad biliproteins – an evolutionary perspective. Photosynth. Res. 46: 93-105
Greenwood AD (1974) The Cryptophyta in relation to phylogeny and photosynthesis. Proc. 8th Int. Congr. Electron Microsc. 2: 566-567
Greenwood AD, Griffiths HB, Santore UJ (1977) Chloroplasts and cell compartments in Cryptophyceae. Brit. Phycol. J. 12: 119
Gould SB, Fan E, Hempel F, Maier UG, Klösgen RB (2007) Translocation of a phycoerythrin α subunit across five biological membranes. J. Biol. Chem. 282: 30295-30302
Gould SB, Waller RF, McFadden GI (2008) Plastid evolution. Annu. Rev. Plant. Biol. 59: 491-517
Hackett JD, Yoon HS, Li S, Reyes-Prieto A, Rummele SE, Bhattacharya D (2007) Phylogenomic analysis supports the monophyly of cryptophytes and haptophytes and the association of rhizaria with chromalveolates. Mol. Biol. Evol. 24: 1702-1713
Hammer A, Schumann R, Schubert H (2002). Light and temperature acclimation of Rhodomonas salina (Cryptophyceae): photosynthetic performance. Aquat. Microb. Ecol. 29: 287-296
Hansgirg A (1885) Anhang zu meiner Abhandlung „Ueber den Polymorphismus der Algen“. Bot. Zentralbl. 23: 229-233
Harper JT, Waanders E, Keeling PJ (2005) On the monophyly of chromalveolates using a six-protein phylogeny of eukaryotes. Int. J. Syst. Evol. Microbiol. 55: 487-496
Hibberd DJ, Greenwood AD, Bronwen Griffiths H (1971) Observations on the ultrastructure of the flagella and periplast in the Cryptophyceae. Br. Phycol. J. 6: 61-72
Hill DRA (1991a) A revised circumscription of Cryptomonas (Cryptophyceae) based on examination of Australian strains. Phycologia 30: 170-188
Hill DRA (1991b) Chroomonas and other blue-green cryptomonads. J. Phycol. 27: 133-145
Hill DRA, Wetherbee R (1986) Proteomonas sulcata gen. et sp. nov. (Cryptophyceae), a cryptomonad with two morphologically distinct and alternating forms. Phycologia 25: 521-543
Hill DRA, Wetherbee R (1988) The structure and taxonomy of Rhinomonas pauca gen. et sp. nov. (Cryptophyceae). Phycologia 27: 355-365
Hill DRA, Wetherbee R (1989) A reappraisal of the genus Rhodomonas (Cryptophyceae). Phycologia 28: 143-158
Hill DRA, Wetherbee R (1990) Guillardia theta gen. et sp. nov. (Cryptophyceae). Can. J. Bot. 68: 1873-1876
Hoef-Emden K (2005) Multiple independent losses of photosynthesis and differing evolutionary rates in the genus Cryptomonas (Cryptophyceae): Combined phylogenetic analyses of DNA sequences of the nuclear and nucleomorph ribosomal operons. J. Mol. Evol. 60: 183-195
Hoef-Emden K (2007) Revision of the genus Cryptomonas (Cryptophyceae) II: Incongruences between classical morphospecies and molecular phylogeny in smaller pyrenoid-less cells. Phycologia 46: 402-428
Hoef-Emden K (2008) Molecular phylogeny of phycocyanin-containing cryptophytes: Evolution of biliproteins and geographical distribution. J. Phycol. 44: 985-993
Hoef-Emden K, Melkonian M (2003) Revision of the genus Cryptomonas (Cryptophyceae): a combination of molecular phylogeny and morphology provides insights into a long-hidden dimorphism. Protist 154: 371-409
Hoef-Emden K, Marin B, Melkonian M (2002) Nuclear and nucleomorph SSU rDNA phylogeny in the Cryptophyta and the evolution of cryptophyte diversity. J. Mol. Evol. 55: 161-179
Huber-Pestalozzi G (1950) Das Phytoplankton des Süßwassers – 3. Teil. Cryptophyceen, Chloromonadinen, Peridineen. E. Schweizerbart'sche Verlagsbuchhandlung, Stuttgart
Javornický P, Hindák F (1970). Cryptomonas frigoris spec. nova (Cryptophyceae), the new cyst-forming flagellate from the snow of the High Tatras. Biologia, Ser. A 25: 241-250
Jenkins J, Hiller RG, Speirs J, Godovac-Zimmermann J (1990) A genomic clone encoding a cryptophyte phycoerythrin a-subunit. Evidence for three a-subunits and an N-terminal membrane transit sequence. FEBS Lett. 273: 191-194
Jezbera J, Horňák K, �imek K (2005) Food selection by bacterivorous protists: Insight from the analysis of the food vacuole content by means of fluorescence in situ hybridization. FEMS Microbiol. Ecol. 52: 351-363
Joyon L (1963) Sur la présence d'un leucoplaste chez la Cryptomonadine decolorée Chilomonas paramecium (Ehrenberg). C. R. Hebd. Séances Acad. Sci. 256: 3502-3503
Karsten G (1898) Rhodomonas baltica N. g. et sp. Wiss. Meeresunters. Abt. Kiel 3: 15
Khan H, Parks N, Kozera C, Curtis BA, Parsons BJ, Bowman S, Archibald JM (2007) Plastid genome sequence of the cryptophyte alga Rhodomonas salina CCMP1319: lateral transfer of putative DNA replication machinery and a test of chromist plastid phylogeny. Mol. Biol. Evol. 24: 1832-1842
Klaveness D (1988). Ecology of the Cryptomonadida: A First Review. In Sandgren CD (ed.) Growth and Reproductive Strategies of Freshwater Phytoplankton. Cambridge University Press, Cambridge, pp 105-133
Kugrens P, Lee RE (1990) Ultrastructural evidence for for bacterial incorporation and myxotrophy in the photosynthetic cryptomonad Chroomonas pochmanni Huber-Pestalozzi (Cryptomonadida). J. Protozool. 37: 263-267
Kugrens P, Lee RE (1991) Organization of cryptomonads. In: The biology of free-living heterotrophic flagellates. Patterson DJ, Larsen J (eds). Clarendon Press, Oxford, p. 219-233
Kugrens P, Lee RE, Andersen RA (1986) Cell form and surface patterns in Chroomonas and Cryptomonas cells (Cryptophyceae) as revealed by scanning electron microscopy. J. Phycol. 22: 512-522
Kugrens P, Lee RE, Andersen RA (1987) Ultrastructural variations in cryptomonad flagella. J. Phycol. 23: 511-518
Lane CE, Archibald JM (2006) Novel nucleomorph genome architecture in the cryptomonad genus Hemiselmis. J. Eukaryot. Microbiol. 44: 339-350
Lane CE, Archibald JM (2008) New marine members of the genus Hemiselmis (Cryptomonadales, Cryptophyceae). J. Phycol. 44: 339-350
Lane CE, Khan H, MacKinnon M, Fong A, Theophilou S, Archibald JM (2006) Insight into the diversity and evolution of the cryptomonad nucleomorph genome. Mol. Biol. Evol. 23: 856-865
Lane CE, van den Heuvel K, Kozera C, Curtis BA, Parsons B, Bowman S, Archibald JM (2007) Nucleomorph genome of Hemiselmis andersenii reveals complete intron loss and compaction as a driver of protein structure and function. Proc. Natl. Acad. Sci. USA 104: 19908-19913
Larsen J, Patterson DJ (1990) Some flagellates (Protista) from tropical marine sediments. J. Nat. Hist. 24: 801-937
Lichtlé C (1979). Effects of nitrogen deficiency and light of high intensity on Cryptomonas rufescens (Cryptophyceae). I. Cell and photosynthetic transformations and encystment. Protoplasma 101: 283-299
Lichtlé C (1980). Effects of nitrogen deficiency and light of high intensity on Cryptomonas rufescens (Cryptophyceae). II. Excystment. Protoplasma 102: 11-19
Maier UG, Hofmann CJ, Eschbach S, Wolters J, Igloi GL (1991) Demonstration of nucleomorph-encoded eukaryotic small subunit ribosomal RNA in cryptomonads. Mol. Gen. Genet. 230: 155-160
Marin B, Klingberg M, Melkonian M (1998) Phylogenetic relationships among the Cryptophyta: analyses of nuclear-encoded SSU rRNA sequences support the monophyly of extant plastid-containing lineages. Protist 149: 265-276
Marshall W, Laybourn-Parry J (2002) The balance between photosynthesis and grazing in Antarctic mixotrophic cryptophytes during summer. Freshw. Biol. 47: 2060-2070
McFadden GI, Gilson PR, Hill DRA (1994) Goniomonas: rRNA sequences indicate that that this phagotrophic flagellate is a close relative to the host component of cryptomonads. Eur. J. Phycol. 29: 29-32
Mignot JP (1965) Étude ultrastructurale de (Cyathomonas truncata) From. (flagellé cryptomonadine). J. Microscopie 4: 239-252
Mignot JP, Joyon L, Pringsheim EG (1968) Compléments a l'étude cytologique des cryptomonadines. Protistologica 4: 493-505
Morrall S, Greenwood AD (1980) A comparison of the periodic substructures of the trichocysts of the Cryptophyceae and Prasinophyceae. BioSystems 12: 71-83
Not F, Valentin K, Romari K, Lovejoy C, Massana R, Tobe K, Vaulot D, Medlin LK (2007) Picobiliphytes: a marine picoplanktonic algal group with unknown affinities to other eukaryotes. Science 315: 253-255
Novarino G (2003) A companion to the identification of cryptomonad flagellates (Cryptophyceae = Cryptomonadea). Hydrobiologia 502: 225-270
Novarino G, Lucas IAN, Morrall S (1994) Observations on the genus Plagioselmis (Cryptophyceae). Cryptogam. Algol. 15: 87-107
Okamoto N, Inouye I (2005) The katablepharids are a distant sister group of the Cryptophyta: A proposal for Katablepharidophyta Divisio Nova/Katablepharida Phylum Novum based on SSU rDNA and beta-tubulin phylogeny. Protist 156: 163-179
Parke M (1949) Studies on marine flagellates. J. Mar. Biol. Ass. UK 28: 255-285
Patron NJ, Inagaki Y, Keeling, PJ (2007) Multiple gene phylogenies support the monophyly of cryptomonad and haptophyte host lineages. Curr. Biol. 17: 887-891
Pastoureaud A, Dupuy, Chrétiennot-Dinet MJ, Lantoine F, Loret P (2003) Red coloration of oysters along the French Atlantic coast during the 1998 winter season: Implication of nanoplanktonic cryptophytes. Aquaculture 228: 225-235
Pedrós-Alió C, Massana R, Latasa M, García-Cantizano J, Gasol JM (1995). Predation by ciliates on a metalimnetic Cryptomonas population – feeding rates, impact and effects of vertical migration. J. Plankton Res. 17: 2131-2154
Pringsheim EG (1968) Zur Kenntnis der Cryptomonaden des Süßwassers. Nova Hedwigia 16: 367-401
Reith M, Douglas S (1990) Localization of a β-phycoerythrin to the thylakoid lumen of Cryptomonas φ does not involve a signal peptide. Plant Mol. Biol. 15: 585-592
Rensing SA, Goddemeier M, Hofmann CJ, Maier UG (1994) The presence of a nucleomorph hsp70 gene is a common feature of Cryptophyta and Chlorarachniophyta. Curr. Genet. 26: 451-455
Salonen K, Jones RI, Arvola L (1984) Hypolimnetic phosphorus retrieval by diel vertical migrations of lake phytoplankton. Freshw. Biol. 14: 431-438
Sanchez-Puerta MV, Bachvaroff TR, Delwiche CF (2007) Sorting wheat from chaff in multi-gene analyses of chlorophyll c-containing plastids. Mol. Phylogenet. Evol. 44: 885-897
Santore UJ (1984) Some aspects of taxonomy in the Cryptophyceae. New Phytol. 98: 627-646
Schnepf E, Melkonian M (1990) Bacteriophage-like particles in endocytic bacteria of Cryptomonas (Cryptophyceae). Phycologia 29: 338-343
Schuster FL (1968) The gullet and trichocysts of Cyathomonas truncata. Exp. Cell Res. 49: 277-284
Sepsenwol S (1973) Leucoplast of the cryptomonad Chilomonas paramecium. Exp. Cell Res. 76: 395-409
Shalchian-Tabrizi K, Eikrem W, Klaveness D, Vaulot D, Minge MA, Le Gall F, Romari K, Throndsen J, Botnen A, Massana R, Thomsen HA, Jakobsen KS (2006) Telonemia, a new protist phylum with affinity to chromist lineages. Proc. R. Soc. Lond. Biol. Sci. 273: 1833-1842
Shalchian-Tabrizi K, Br�te J, Logares R, Klaveness D, Berney C, Jakobsen KS (2008) Diversification of unicellular eukaryotes: cryptomonad colonizations of marine and fresh waters inferred from revised 18S rRNA phylogeny. Environ. Microbiol. 10: 2635-2644
Skuja H (1948) Taxonomie des Phytoplanktons einiger Seen in Uppland, Schweden. Symb. Bot. Ups. 9: 346-367
Sommer MS, Gould SB, Lehmann P, Gruber A, Przyborski JM, Maier UG (2007) Der1-mediated preprotein import into the periplast compartment of chromalveolates? Mol. Biol. Evol. 24: 918-928
Starmach K (1974) Flora Słodkowodna Polski. Tom 4. Cryptophyceae – Kryptofity, Dinophyceae – Dinofity, Raphidophyceae – Rafidofity. Państwowe Wydawnictwo Naukowe, Warszawa – Krakow
Stein F Ritter von (1878) Der Organismus der Infusionsthiere. W. Engelmann, Leipzig. Vol. 3.1
Tirok K, Gaedke U (2007). Regulation of planktonic ciliate dynamics and functional composition during spring in Lake Constance. Aquat. Microb. Ecol. 49: 87-100
Tranvik LJ, Porter KG, Sieburth JMcN (1989) Occurrence of bacterivory in Cryptomonas, a common freshwater phytoplankter. Oecologia 78: 473-476
Vesk M, Dwarte D, Fowler S, Hiller RG (1992) Freeze fracture immunocytochemistry of light-harvesting pigment complexes in a cryptophyte. Protoplasma 170: 166-176
von der Heyden S, Chao E, Cavalier-Smith T (2004) Genetic diversity of goniomonads: An ancient divergence between marine and freshwater species. Eur. J. Phycol. 39: 343-350
Wilk KE, Harrop SJ, Jankova L, Edler D, Keenan G, Sharples F, Hiller RG, Curmi PMG (1999) Evolution of a light-harvesting protein by addition of new subunits and rearrangement of conserved elements: Crystal structure of a cryptophyte phycoerythrin at 1.63-Å resolution. Proc. Natl. Acad. Sci. USA 96: 8901-8906
Williams BA, Slamovits CH, Patron NJ, Fast NM, Keeling PJ (2005) A high frequency of overlapping gene expression in compacted eukaryotic genomes. Proc. Natl. Acad. Sci. USA 102: 10936-10941
Yoon HS, Hackett JD, Pinto G, Bhattacharya D (2002) The single, ancient origin of chromist plastids. Proc. Natl. Acad. Sci. USA 99: 15507-15512
Title Illustrations

| Identified By | Kerstin Hoef-Emden |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2008 Kerstin Hoef-Emden

|
| Scientific Name | Rhodomonas sp. |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2008 Kerstin Hoef-Emden

|
| Scientific Name | Chroomonas sp. palmella |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 2008 Kerstin Hoef-Emden

|
About This Page
This page is being developed as part of the Tree of Life Web Project Protist Diversity Workshop, co-sponsored by the Canadian Institute for Advanced Research (CIFAR) program in Integrated Microbial Biodiversity and the Tula Foundation.
Kerstin Hoef-Emden

Universit�t zu K�ln, K�ln, Germany
John M. Archibald

Dalhousie University, Halifax, Nova Scotia, Canada
Correspondence regarding this page should be directed to Kerstin Hoef-Emden at and John M. Archibald at
Page copyright © 2010 Kerstin Hoef-Emden and John M. Archibald
 Page: Tree of Life
Cryptomonads = Goniomonas + plastid-containing cryptophytes. Cryptophyta.
Authored by
Kerstin Hoef-Emden and John M. Archibald.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Cryptomonads = Goniomonas + plastid-containing cryptophytes. Cryptophyta.
Authored by
Kerstin Hoef-Emden and John M. Archibald.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 14 September 2008
- Content changed 02 April 2010
Citing this page:
Hoef-Emden, Kerstin and John M. Archibald. 2010. Cryptomonads = Goniomonas + plastid-containing cryptophytes. Cryptophyta. Version 02 April 2010. http://tolweb.org/Cryptomonads/2396/2010.04.02 in The Tree of Life Web Project, http://tolweb.org/














 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site