Sodium oxide is a chemical compound with the formula Na2O. It is used in ceramics and glasses. It is a white solid but the compound is rarely encountered. Instead "sodium oxide" is used to describe components of various materials such as glasses and fertilizers which contain oxides that include sodium and other elements. Sodium oxide is a component.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sodium oxide
| |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ECHA InfoCard | 100.013.827 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| UN number | 1825 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| Na2O | |||
| Molar mass | 61.979 g·mol−1 | ||
| Appearance | white solid | ||
| Density | 2.49 g/cm3 | ||
| Melting point | 1,132 °C (2,070 °F; 1,405 K) | ||
| Boiling point | 1,950 °C (3,540 °F; 2,220 K) sublimates | ||
| sublimates at 1275 °C | |||
| Reacts to form NaOH | |||
| Solubility | Reacts with ethanol | ||
| −19.8·10−6 cm3/mol | |||
| Structure | |||
| Antifluorite (face centered cubic), cF12 | |||
| Fm3m, No. 225 | |||
| Tetrahedral (Na+); cubic (O2−) | |||
| Thermochemistry | |||
Heat capacity (C)
|
72.95 J/(mol·K) | ||
Std molar
entropy (S⦵298) |
73 J/(mol·K)[1] | ||
Std enthalpy of
formation (ΔfH⦵298) |
−416 kJ/mol[1] | ||
Gibbs free energy (ΔfG⦵)
|
−377.1 kJ/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
corrosive, reacts violently with water | ||
| GHS labelling: | |||
 [2] [2]
| |||
| H314 | |||
| P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | nonflammable | ||
| Safety data sheet (SDS) | ICSC 1653 | ||
| Related compounds | |||
Other anions
|
|||
Other cations
|
|||
Related compounds
|
Sodium hydroxide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
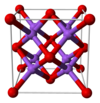
Structure
editThe structure of sodium oxide has been determined by X-ray crystallography. Most alkali metal oxides M2O (M = Li, Na, K, Rb) crystallise in the antifluorite structure. In this motif the positions of the anions and cations are reversed relative to their positions in CaF2, with sodium ions tetrahedrally coordinated to 4 oxide ions and oxide cubically coordinated to 8 sodium ions.[3][4]
Preparation
editSodium oxide is produced by the reaction of sodium with sodium hydroxide, sodium peroxide, or sodium nitrite:[5]
- 2 NaOH + 2 Na → 2 Na2O + H2
To the extent that NaOH is contaminated with water, correspondingly greater amounts of sodium are employed. Excess sodium is distilled from the crude product.[6]
A second method involves heating a mixture of sodium azide and sodium nitrate:[6]
- 5 NaN3 + NaNO3 → 3 Na2O + 8 N2
Burning sodium in air produces a mixture of Na2O and sodium peroxide (Na2O2).
A third much less known method involves heating sodium metal with iron(III) oxide (rust):
- 6 Na + Fe2O3 → 3 Na2O + 2 Fe
the reaction should be done in an inert atmosphere to avoid the reaction of sodium with the air instead.
Applications
editGlassmaking
editGlasses are often described in terms of their sodium oxide content although they do not really contain Na2O. Furthermore, such glasses are not made from sodium oxide, but the equivalent of Na2O is added in the form of "soda" (sodium carbonate), which loses carbon dioxide at high temperatures:
A typical manufactured glass contains around 15% sodium oxide, 70% silica (silicon dioxide), and 9% lime (calcium oxide). The sodium carbonate "soda" serves as a flux to lower the temperature at which the silica mixture melts. Such soda-lime glass has a much lower melting temperature than pure silica and has slightly higher elasticity. These changes arise because the Na2[SiO2]x[SiO3]-based material is somewhat more flexible.
Reactions
editSodium oxide reacts readily and irreversibly with water to give sodium hydroxide:
- Na2O + H2O → 2 NaOH
Because of this reaction, sodium oxide is sometimes referred to as the base anhydride of sodium hydroxide (more archaically, "anhydride of caustic soda").
References
edit- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A23. ISBN 978-0-618-94690-7.
- ^ Sigma-Aldrich Co., Sodium oxide. Retrieved on 2014-05-25.
- ^ Zintl, E.; Harder, A.; Dauth B. (1934). "Gitterstruktur der oxyde, sulfide, selenide und telluride des lithiums, natriums und kaliums". Zeitschrift für Elektrochemie und Angewandte Physikalische Chemie. 40 (8): 588–93. doi:10.1002/bbpc.19340400811. S2CID 94213844.
- ^ Wells, A. F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press ISBN 0-19-855370-6
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ a b E. Dönges (1963). "Sodium Oxide (IV)". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 1pages=975-6. NY, NY: Academic Press.